I’ve talked about dyeing with madder more than a few times. I’ve written up documentation about some of my early madder experiments. I don’t think anyone would be surprised to know that I am really rather fond of playing with madder. So let’s do a little bit of a wander down the botanical path with the plant.
NB. I am a terrible gardener. I managed to kill a mint plant, that’s the level of terrible we are talking here. I do not grow my own. I let professionals (or awesome friends) do that for me and reap the bounty of their labours with gratitude (and/or payment.)

Right, we’re talking about Madder. Most commonly, when the word madder is tossed around in the dye world, folks are meaning the dried and ground roots of Rubia tinctorum. Also referred to as common madder or European madder. It is, however, not the only madder plant out there that has dyestuff for us! It’s not even the only madder plant we can buy. The other one that’s easy to find at the dye supplier is Rubia cordifolia. Also referred to as Indian madder, or munjeet. But yet again that’s not the only other madder out there, there’s more! Wild madder, Rubia peregrina and Japanese madder, Rubia akane and there are others in the Rubia family.. but the first two are the most commonly used.
R. tinctorum is a perennial that was cultivated throughout Europe and the Middle East, with the highest quality coming from Turkey, Holland and France. It is native to western and central Asia and naturalized itself in central and southern Europe. Munjeet (R. cordifolia) comes from moutainous regions of Asia, from the Himalayas to Japan and also in tropical Africa. Wild madder (R. peregrina) is a native of Europe, Turkey and North Africa as well as the coastal regions of southern England and some bits of Wales and Ireland. The roots of wild madder are smaller than that of madder, requiring more dye stuff to gain a strong colour than one would require from madder, although that can be mitigated by waiting longer to harvest the wild madder roots (five years, rather than three for R. tinctorum)

All of the various versions of madder have been used since antiquity, with evidence in extant items from Mohenjo-Daro in the Indus vally (approx 3000 BCE) and mentions in Pliny the Elder’s Naturalis Historia and recipes using madder are found in the Papyrus Graecus Holmiensis (approx 4th century) Assumptions about which madder was being used are usually based on geography. The East was more likely to be using R. cordifolia, the West more likely to be using R. tinctorum or R. peregrina. Local traditions using local plants, as the Rubia plant family is happy to grow in so very many different places.
All of them contain similar dye molecules, although different plant species have them in different combinations and concentrations. This chart from “The Colourful Past” by Judith H. Hofenk de Graaff is an excellent summary of who has what, with some extra plants that also contain anthraquinones.

Forgive me for being the kind of person who casually drops words like ‘anthraquinones’ in conversation, but in a nutshell.. those are the dye molecules that provide the red colour. For the curious, these are their structures (why this graphic is missing alizarin, I have no idea but it’s below):
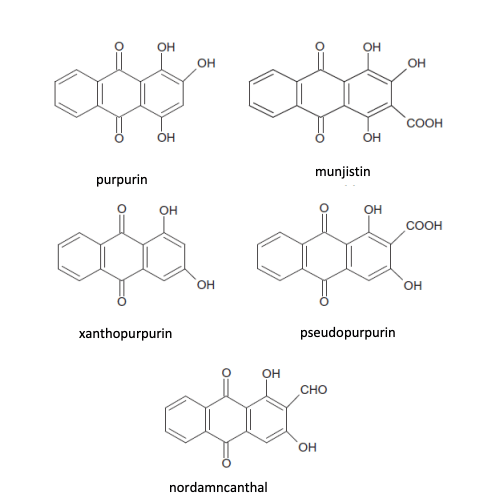

Phew, okay.. still with me after the brief foray into chemistry? (If you’re interested, the handout for my class on dye molecules can be found here. If you have a virtual event you’d like me to teach it at, just ping me.)
At the end of the day, if you have madder, munjeet or wild madder, you’re getting a dose of very similar dye molecules and just enjoy the ride. All of them function pretty similarly, and the reds are just so much fun to work with. I have an experiment in progress comparing R. tinctorum with R. cordifolia, through a collection of exhaust baths, so look for that coming soon!
Bibliography:
Hofenk de Graaff, Judith H., Wilma G. Th Roelofs, and Maarten R. van Bommel. The Colourful Past: Origins, Chemistry and Identification of Natural Dyestuffs. London: Archetype Publ, 2004.
Dean, Jenny. Wild Color: The Complete Guide to Making and Using Natural Dyes. Rev. and Updated ed., 1st rev. U.S. ed. New York: Watson-Guptill, 2010.
Cannon, John, and Gretel Dalby-Quenet, eds. Dye Plants and Dyeing. Repr. London: Black, 2002.
Yusuf, Mohd, Mohammad Shahid, Shafat Khan, Mohd Khan, Shahid Salam, Faqeer Mohammad, and Mohd Khan. “Eco-Dyeing of Wool Using Aqueous Extract of the Roots of Indian Madder (Rubia Cordifolia) as Natural Dye.” Journal of Natural Fibers 10 (March 13, 2013): 14–18. https://doi.org/10.1080/15440478.2012.738026.
Kauffman, George B., ed. Coordination Chemistry: A Century of Progress. Vol. 565. ACS Symposium Series. Washington, DC: American Chemical Society, 1994. https://doi.org/10.1021/bk-1994-0565.
